
Prof Kana M. Sureshan
Professor (Chemistry)
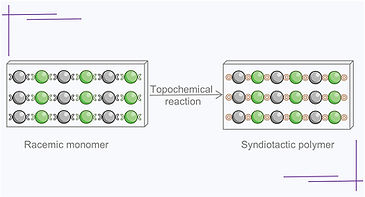
136. Topochemical Synthesis of a Syndiotactic Polymer from a Racemic Monomer
S. K. Gupta, Dr. R. Khazeber, K.S. Siddharth, A. Balakrishnan and Prof. Dr. K. M. Sureshan*
J. Am. Chem. Soc., https://doi.org/10.1021/jacs.5c18292
135. Topochemical Alkyne Nitrile Oxide Cycloaddition for Polymer Synthesis
Dr. R. Singh, A. Siddhaarthan and Prof. Dr. K. M. Sureshan*
Angew. Chem. Int. Ed., https://doi.org/10.1002/anie.202520947

134. Single-Crystal-to-Single-Crystal Synthesis of an Adaptive Two-Dimensional Polymer with Dynamic Pores
H. Balan, S. Shahanas and Prof. Dr. K. M. Sureshan*
J. Am. Chem. Soc., https://doi.org/10.1021/jacs.5c13942

133. Topochemical Polymerization of Retro-Isomeric Peptides for Tuning the Polymer Structure and Properties
A. Lal, R. Rai, M.C. Madhusudhanan and Prof. Dr. K. M. Sureshan*
Angew. Chem. Int. Ed., https://doi.org/10.1002/anie.202503624

132. Isomer-Dependent Reactivity in the Solid State: Topochemical [4+4] vs. [4+2] Cycloaddition Reactions
A. Lal and Prof. Dr. K. M. Sureshan*
Chem. Sci., https://doi.org/10.1039/D5SC02327K
.png)
131. Absolute Asymmetric Synthesis of a Homochiral Polymer from an Achiral Monomer
D. Xavier, S. Pathak, Dr. C. R. Göb and Prof. Dr. K. M. Sureshan*
Angew. Chem. Int. Ed., https://doi.org/10.1002/anie.202510058

130. Single-Crystal-to-Single-Crystal Synthesis of a Rope-Ladder Polymer
H. Balan, G. Sadasivan, E. Paul and Prof. Dr. K. M. Sureshan*
Angew. Chem. Int. Ed., https://doi.org/10.1002/anie.202506699

129. Supramolecular Preorganization of Amine-functionalized Diacetylene Monomers in their Crystals Allows their Topochemical Polymerization to Polydiacetylenes Capable of CO2 Capture
A. Lal, Dr. C. Raju and Prof. Dr. K. M. Sureshan*
Chem. Eur. J. 2025, 31, e202403935

128. Single-Crystal-to-Single-Crystal Synthesis of a Polymer in Two Distinct Topologies
Dr. J. R. Pathan, H. Balan, K. Das, Prof. Dr. C. M. Reddy and Prof. Dr. K. M. Sureshan*
Angew. Chem. Int. Ed., https://doi.org/10.1002/anie.202500646

127. A Malleable Collagen-Mimic that Undergoes Moisture-Induced Hardening for Gluing Hydrophilic Surfaces
R. Rai, D. Xavier, S. Pathak, F. B. Fernandez, M. Komath, K. M. Sureshan*
Angew. Chem. Int. Ed., 2025, 64, e202422593
126. Light-induced transformation of a supramolecular gel to a stronger covalent polymeric gel
S. K. Saleem, T. Pramod, P. Kuruva, S. V. Haridas, A. Shanmugam, M. Thalakulam, K. M. Sureshan*

125. A syndiotactic polymer via spontaneous exo-selective single-crystal-to-single-crystal topochemical Diels-Alder cycloaddition reaction
S. Pathak, K. M. Sureshan*
J. Am. Chem. Soc. 2024, 146, 30495-30501.

124. Unclicking the click: A depolymerizable clicked polymer via two consecutive single-crystal-to-single-crystal reactions
B. Sebastian, K. M. Sureshan*
Angew. Chem. Int. Ed., 2024, 63, e202417905

123. A self-healing crystal that repairs multiple cracks
J. R. Pathan, H. Balan, P. Commins, A. Ravi, M. B. Al-Handawi, I. C.-Y. Hou, P. Naumov, K. M. Sureshan*
J. Am. Chem. Soc. 2024, 146, 27100-27108.

122. Hierarchical single-crystal-to-single-crystal transformations of a monomer to a 1D-polymer and then to a 2D-polymer
H. Balan, K. M. Sureshan*

121. Simultaneous and in situ syntheses of an enantiomeric pair of homochiral polymers as their perfect stereocomplex in a crystal
R. Khazeber, S. Pathak, K. M. Sureshan*

120. Large molecular rotation in crystal changes the course of a topochemical Diels-Alder reaction from a predicted polymerization to an unexpected intramolecular cyclization
A. Lal, M. C. Madhusudhanan, K. M. Sureshan*
Angew. Chem. Int. Ed., 2024, 63, e202411165.

119. Massive molecular motion in crystal leads to an unexpected helical covalent polymer in a solid-state polymerization
R. Khazeber, G. S. Kana, K. M. Sureshan*

118. Topochemistry for difficult peptide-polymer synthesis: Single-crystal-to-single-crystal synthesis of an isoleucine-based polymer, a hydrophobic coating material
T. Pramod, R. Khazeber, V. Athiyarath, K. M. Sureshan*
J. Am. Chem. Soc. 2024, 146, 7257-7265.

117. Single-crystal-to-single-crystal topochemical synthesis of a collagen-inspired covalent helical polymer
R. Rai, R. Khazeber, K. M. Sureshan*
Angew. Chem. Int. Ed., 2023, 62, e202315742

116. Two structurally different polymers from a single monomer
J. R. Pathan, S. Bhandary, K. M. Sureshan*
J. Am. Chem. Soc. 2023, 145, 22633-22638.

115. Topochemical syntheses of polyarylopeptides involving large molecular motions: frustrated monomer packing leads to the formation of polymer blends
C. Raju, K. Mridula, N. Srinivasan, S. Kunnikuruvan, K. M. Sureshan*
Angew. Chem. Int. Ed., 2023, 62, e202306504.

114. Adamantoid scaffolds for multiple cargo loading and cellular delivery as β-cyclodextrin inclusion complexes
A. Ravi, A. Pathigoolla, H. Balan, R. Gupta, G. Raj, R. Varghese, K. M. Sureshan*
Angew. Chem. Int. Ed., 2023, 62, e202307324.

113. Rational Design and Topochemical Synthesis of Polymorphs of a Polymer
V. Athiyarath, L. A. Mathew, Y. Zhao, R. Khazeber, U. Ramamurty, K. M. Sureshan*
112. Regiospecific synthesis of a reprocessable galactan-mimic via topochemical polymerization
A. Ravi, S. Z. Hassan, A. Pathigoolla, A. Lal, B. Varghese, K. M. Sureshan*
ACS Sust. Chem. Engg. 2023, 11, 7210–7217

111. Cascading effect of large molecular motion in crystals: A topotactic polymorphic transition paves the way to topochemical polymerization
C. Raju, G. R. Ramteke, K. V. J. Jose, K. M. Sureshan*
J. Am. Chem. Soc. 2023 DOI: 10.1021/jacs.3c00132

110. Tuning the Regioselectivity of Topochemical Polymerization through Cocrystallization of the Monomer with an Inert Isostere
K. Hema, C. Raju, S. Bhandary, K. M. Sureshan*
Angew. Chem. Int. Ed., https://doi.org/10.1002/anie.202210733
_tif.png)
109. Topochemical Cycloaddition Reaction between an Azide and an Internal Alkyne
C. Raju, S. Kunnikuruvan, K. M. Sureshan*
Angew. Chem. Int. Ed., https://doi.org/10.1002/anie.202210453

108. Single-crystal-to-single-crystal translation of a helical supramolecular polymer to helical covalent polymer
R. Khazeber, K. M. Sureshan*
PNAS, https://doi.org/10.1073/pnas.2205320119
107. A Biomaterial-Based Porous Core–Shell Sorbent for Practical and Efficient Marine Oil Spill Recovery
C. Raju, L. A. Mathew, K. M. Sureshan*
Adv. Sustainable Syst., https://doi.org/10.1002/adsu.202100521

106. Azide···alkyne interaction: A crucial attractive force for their preorganization for topochemical cycloaddition reaction
S. Bhandary,A. Pathigoolla,M. C. Madhusudhanan, K. M. Sureshan*
Chem. Eur. J., 2022, https://doi.org/10.1002/chem.202200820
105.Topochemical Postulates: Are They Relevant for Topochemical Reactions Occurring at Elevated Temperatures?
A. Ravi, Syed Z. Hassan ,S. Bhandary ,K. M. Sureshan*
Angew. Chem. Int. Ed., 2022, https://doi.org/10.1002/ange.202200954

104. Topochemical Synthesis of a Heterochiral Peptide Polymer in Different Polymorphic Forms from Crystals and Aerogels
R. Rai, K. M. Sureshan*
Angew. Chem. Int. Ed., 2022, https://doi.org/10.1002/anie.202111623
103. Secondary Structure Tuning of a Pseudoprotein Between β-Meander and α-Helical Forms in the Solid-State
V. Athiyarath, M. C. Madhusudhanan, S. Kunnikuruvan,, K. M. Sureshan*
Angew. Chem. Int. Ed., 2021, https://doi.org/10.1002/anie.202113129

102. Azide···Oxygen Interaction: A Crystal Engineering Tool for Conformational Locking
M. C. Madhusudhanan, H. Balan, D. B. Werz, K. M. Sureshan*
Angew. Chem. Int. Ed., 2021, 60, 22797-22803

101. Topochemical ene-azide cycloaddition reaction
R. Khazeber, K. M. Sureshan*
Angew. Chem. Int. Ed., 2021, 60, 24875-24881

100. Single-crystal-to-single-crystal synthesis of a pseudostarch via topochemical azide–alkyne cycloaddition polymerization
A. Ravi, A. Shijad, K. M. Sureshan*
Chem. Sci., 2021, 12, 11652-11658

99. Solvent-free and catalyst-free synthesis of cross-linkable polyfumaramides via topochemical azide-alkyne cycloaddition polymerization
J. R. Pathan, K. M. Sureshan*
ACS Sustainable Chem. Eng. 2021, 9, 9871–9878

98. Novel Substrates for Kinases involved in the biosynthesis of inositol pyrophosphates and their enhancement of ATPase activity of a kinase
R. Mohanrao, R. Manorama, S. Ganguli, M. C. Madhusudhanan, R. Bhandari, K. M. Sureshan*

97. Synthesis of novel seven-membered carbasugars and evaluation of their glycosidase inhibition potentials.
V. Athiyarath, N. J. Roy, A. T. V. Vijil, K. M. Sureshan*
RSC Adv., 2021, 11, 9410-9420.

96. Polymers with advanced structural and supramolecular features through
topochemical polymerization.
K. Hema, A. Ravi, C. Raju, K. M. Sureshan*
Chem. Sci., 2021, 12, 5361-5380.

95. Topochemical polymerizations for the solid-state synthesis of organic polymers.
K. Hema, A. Ravi, C. Raju, J. R. Pathan, R. Rai, K. M. Sureshan*
Chem. Soc. Rev., 2021, 50, 4062-4099.

93. Quantification of non‐covalent interactions in azide‐pnictogen, ‐chalcogen, and ‐halogen contacts.
M. Bursch, L. Kunze, A. M. Vibhute, A. Hansen, K. M. Sureshan, P. G. Jones, S. Grimme, D. B Werz*
Chem. Eur. J., 2021, 27, 4627-4639.

2020
94. Scalable topochemical synthesis of a pseudoprotein in aerogel for water-capturing applications.
R. Mohanrao, K. Hema, K. M. Sureshan*
ACS Appl. Polym. Mater., 2020, 2, 11, 4985-4992.
92. How far are we in combating marine oil spills using phase selective organogelators?
A. M. Vibhute, K. M. Sureshan*
ChemSusChem., 2020, 13, 5343-5360

91. Designed synthesis of a 1D polymer in twist‐stacked topology via single‐crystal‐to‐single‐crystal polymerization.
V. Athiyarath, K. M. Sureshan*
Angew. Chem. Int. Ed., 2020, 59, 15580-15585

90. β‐Sheet to Helical‐sheet Evolution Induced by Topochemical Polymerization: Cross‐α‐amyloid‐like Packing in a Pseudoprotein with Gly‐Phe‐Gly Repeats
K. Hema, K. M. Sureshan*
Angew. Chem. Int. Ed., doi.org/10.1002/anie.201914975
89. Topochemical synthesis of different polymorphs of polymers as a paradigm for tuning properties of polymers
R. Mohanrao, K. Hema, K. M. Sureshan*
Nat. Commun., 2020, doi.org/10.1038/s41467-020-14733-y
88. Crystal‐to‐crystal synthesis of helically ordered polymers of trehalose via topochemical polymerization
K. Hema, R. G. Gonnade, K. M. Sureshan*
Angew. Chem. Int. Ed., 2020, 59, 2897-2903.
2019
87. Topochemical Azide–Alkyne Cycloaddition Reaction
K. Hema, K. M. Sureshan*
86. Halobenzyl alcohols as structurally simple gelators.
A. Prathap, A. Ravi, J. Pathan, K. M. Sureshan*
CrystEngComm, 2019, 21, 5310-5316.
- Sugar-based organogelators for various applications.
A. Prathap, K. M. Sureshan*
Langmuir, 2019, 35, 6005-6014. (Invited perspective) - Solid‐state synthesis of two different polymers in a single crystal: A miscible polymer blend from topochemical reaction.
K. Hema, K. M. Sureshan*
Angew. Chem. Int. Ed., 2019, 58, 2754-2759. - Spontaneous single-crystal-to-single-crystal evolution of two cross-laminated polymers.
V. Athiyarath, K. M. Sureshan*
Angew. Chem. Int. Ed., 2019, 58, 612-617.
2018
- Synthesis and reversible hydration of a pseudoprotein, a fully organic polymeric desiccant by multiple single‐crystal‐to‐single‐crystal transformations.
R. Mohan Rao, K. M. Sureshan*
Angew. Chem. Int. Ed., 2018, 57, 12435-12439. - Tunable mechanical response from a crystal undergoing topochemical dimerization: Instant explosion at faster rate and chemical storage of ‘harvestable explosion’ at slower rate.
A. Ravi, K. M. Sureshan*
Angew. Chem. Int. Ed., 2018, 57, 9362-9366. - Organogel-derived covalent-noncovalent hybrid polymers as alkali metal ion scavengers for partial deionization of water.
A. Prathap, C. Raju, K. M. Sureshan*
ACS Appl. Mater. Interfaces, 2018, 10, 15183-15188. - Model molecules to classify CH∙∙∙O hydrogen-bonds.
A. M. Vibhute, U. D. Priyakumar, A. Ravi, K. M. Sureshan*
Chem. Commun., 2018, 54, 4629-4632. - Chirality-controlled spontaneous twisting of crystals due to thermal topochemical reaction.
R. Rai, B. P. Krishnan, K. M. Sureshan*
Proc. Natl. Acad. Sci. U.S.A. 2018, 115, 2896-2901. - Three-way competition in a topochemical reaction: Permutative azide-alkyne cycloaddition reactions leading to a vast library of products in the crystal.
K. Hema, K. M. Sureshan*
CrystEngComm, 2018, 20, 1478-1482. - A library of multi-purpose supramolecular supergelators: fabrication of structured silica, porous plastics, and fluorescent gel.
B. P. Krishnan, K. M. Sureshan*
Chem. Asian J. 2018, 13, 187-193.
2017
- Organogelator-cellulose composite for practical and eco-friendly marine oil spill recovery.
A. Prathap, K. M. Sureshan*
Angew. Chem. Int. Ed., 2017, 56, 9405 – 9409. - SN2 reactions for rapid syntheses of azido-inositols by one-pot sequence-specific nucleophilysis.
A. Ravi, S. Z. Hassan, A. N. Vanikrishna, K. M. Sureshan*
Chem. Commun., 2017, 53, 3971-3973. - Topochemical azide-alkyne cycloaddition reaction in gels: Size-tunable synthesis of triazole-linked polypeptides.
B. P. Krishnan, K. M. Sureshan*
J. Am. Chem. Soc. 2017, 139, 1584−1589.
2016
- Organogel-assisted topochemical synthesis of multivalent glyco-polymer for high-affinity lectin binding.
B. P. Krishnan, S. Raghu, S. Mukherjee, K. M. Sureshan*
Chem. Commun. 2016, 52, 14089-14092. (Highlighted as cover article). - Carbasugar synthesis via vinylogous ketal: Total syntheses of (+)-MK7607, (-)-MK7607, (-)-Gabosine A, (-)-Epoxydine B, (-)-Epoxydine C, epi-(+)-Gabosine E and epi-(+)-MK7607.
S. Mondal, K. M. Sureshan*
J. Org. Chem., 2016, 81, 11635−11645. - Crystal-to-crystal synthesis of triazole-linked pseudo-proteins via topochemical azide–alkyne cycloaddition reaction.
B. P. Krishnan, R. Rai, A. Ashokan, K. M. Sureshan*
J. Am. Chem. Soc., 2016, 138, 14824−14827. - Synthesis of dimeric analogs of Adenophostin A that potently evoke Ca2+ release through IP3 receptors.
A. M. Vibhute, P. Pushpanandan, M. Varghese, V. Koniecnzy, C. W. Taylor, K. M. Sureshan*
RSC Adv., 2016, 6, 86346-86351. - A molecular level study of metamorphosis and strengthening of gel via spontaneous polymorphic transition.
B. P. Krishnan, K. M. Sureshan*
ChemPhysChem., 2016, 17, 3062-3067. - A versatile glycosylation strategy via Au(III) catalyzed activation of thioglycoside donors.
A. M. Vibhute, A. Dhaka, A. Vignesh, K. M. Sureshan*
Chem. Sci., 2016, 7, 4259-4263. - A sugar-based gelator for marine oil-spill recovery.
A. M. Vibhute, V. Muvvala, K. M. Sureshan*
Angew. Chem. Int. Ed., 2016, 55, 7782-7785. - CaO nanocrystals grown over SiO2 microtubes for efficient CO2 capture: Organogel sets the platform.
A. Prathap, M. M. Shaijumon, K. M. Sureshan*
Chem. Commun. 2016, 52, 1342-1345. - Topochemical Synthesis of triazole-linked homobasic DNA.
A. Pathigoolla, K. M. Sureshan*
Chem. Commun., 2016, 52, 886-888. - Semi-conducting fabrics via in situ topochemical synthesis of polydiacetylene: A new dimension to the use of organogels.
B. P. Krishnan, S. Mukherjee, P. M. Aneesh, M. A. G. Namboothiry, K. M. Sureshan*
Angew. Chem. Int. Ed., 2016, 55, 2345–2349.
2015
- Stoichiometric sensing to opt between gelation and crystallization.
A. Vidyasagar, K. M. Sureshan*
Angew. Chem. Int. Ed., 2015, 54, 12078-12082. - Triazolophostins: A library of novel and potent agonists of IP3 receptors.
A. M. Vibhute, K. M. Sureshan*
Org. Biomol. Chem., 2015, 13, 6698-6710. - Total syntheses of five Uvacalols: Structural validation of Uvacalol A, Uvacalol B and Uvacalol C and disproval of the structures of Uvacalol E and Uvacalol G.
A. Vidyasagar, K. M. Sureshan*
Org. Biomol. Chem., 2015, 13, 3900. - A spontaneous single-crystal-to-single-crystal polymorphic transition involving major packing changes.
B. P. Krishnan, K. M. Sureshan*
J. Am. Chem. Soc., 2015, 137, 1692-1696.
2014
- Hopping mediated anion transport through a mannitol-based rosette ion channel.
T. Saha, S. Dasari, D. Tewari, A. Prathap, K. M. Sureshan, A. K. Bera, A. Mukherjee, P. Talukdar
J. Am. Chem. Soc., 2014, 136, 14128–14135. - Total syntheses and structural Validation of Lincitol A, Lincitol B, Uvacalol I, Uvacalol J and Uvacalol K.
S. Mondal, K. M. Sureshan*
Org. Biomol. Chem., 2014, 12, 7279–7289. - Synthesis of triazole-linked homonucleoside polymers through topochemical azide–alkyne cycloaddition.
A. Pathigoolla, K. M. Sureshan*
Angew. Chem. Int. Ed. 2014, 53, 9522-9525. - Strength from weakness: Conformational divergence between solid and solution states of substituted cyclitols facilitated by CH···O hydrogen bonding.
A. M. Vibhute, K. M. Sureshan*
J. Org. Chem., 2014, 79, 4892-4908. - Bio-inspired synthesis of rare and unnatural carbohydrates and cyclitols through strain driven epimerization.
R. Mohnarao, A. Asokan, K. M. Sureshan*
Chem. Commun. 50, 2014, 50, 6707-6710. - Reverse-CD mimics with flexible linkages offer adaptable cavity sizes for guest encapsulation.
A. Pathigoolla, K. M. Sureshan*
Chem. Commun., 2014, 50, 317-319. - Total synthesis and glycosidase inhibition studies of (–)-Gabosine J and its derivatives.
A. Vidyasagar, K. M. Sureshan*
Eur. J. Org. Chem., 2014, 2349-2356.
2013
- Vinylogy in orthoester hydrolysis: Total syntheses of Cyclophellitol, Valienamine, Gabosine K, Valienone, Gabosine G, 1-epi-streptol, Streptol, and Uvamalol A.
S. Mondal, A. Prathap, K. M. Sureshan*
J. Org. Chem., 2013, 78, 7690-7700. - Chemoselective alcoholysis/acetolysis of trans-ketals over cis-ketals and its application in the total synthesis of the cellular second messenger, D-myo-inositol 1,4,5-trisphosphate.
A. Vidyasagar, A. Pathigoolla, K. M. Sureshan*
Org. Biomol. Chem. 2013, 11, 5443-5453. - A crystal-to-crystal synthesis of triazolyl linked polysaccharide.
A. Pathigoolla, K. M. Sureshan*
Angew. Chem. Int. Ed. 2013, 52, 8671-8675. - H2SO4-silica: An eco-friendly heterogeneous catalyst for the differential protection of myo-inositol hydroxyl groups.
A. M. Vibhute, K. M. Sureshan*
RSC Adv. 2013, 3, 7321-7329. - Weak becomes strong: Remarkable strength of CH...Pi hydrogen bond in the presence of OH...O hydrogen bonds in the crystal stabilization.
K. M. Sureshan*, R. G. Gonnade
CrystEngComm, 2013, 15, 1676-1679. - A versatile solvent-free azide-alkyne click reaction catalyzed by in situ generated copper nanoparticles.
A. Pathigoolla, R. P. Pola, K. M. Sureshan*
Appl. Catal. A: General 2013, 453, 151-158. - Supramolecular design of a bicomponent topochemical reaction between two non-identical molecules.
B. P. Krishnan, S. Ramakrishnan, K. M. Sureshan*
Chem. Commun., 2013, 49, 1494-1496.
2012
- Topochemical click reaction: Spontaneous self-stitching of a monosaccharide to linear oligomers through lattice controlled azide-alkyne cycloaddition.
P. Atchutarao, R. G. Gonnade, K. M. Sureshan*
Angew. Chem. Int. Ed., 2012, 51, 4362-4366. - Mannitol based phase selective supergelator offers a simple, viable and greener method to combat marine oil spills.
A. Prathap, K. M. Sureshan*
Chem. Commun., 2012, 48, 5250-5252. - Contribution of phosphates and adenine to the potency of adenophostins at the IP3 receptor: Synthesis of all possible bisphosphates of Adenophostin A.
K. M. Sureshan, A. M. Riley, M. Thomas, S. Tovey, C. W. Taylor, B. V. L. Potter
J. Med. Chem., 2012, 55, 1706-1720. - Regioselectivity among six secondary hydroxyl groups: Selective acylation of the least reactive hydroxyl groups of inositol.
A. M. Vibhute, A. Vidyasagar, S. Sarala, K.M. Sureshan*
Chem. Commun., 2012, 48, 2448-2450. - Strength from weakness: Opportunistic CH...O interactions differentially dictate the conformational fate in solid and solution states.
A. M. Vibhute, R. G, Gonnade, R.S. Swathi, K. M. Sureshan*
Chem. Commun., 2012, 48, 717-719. - Strength from weakness: The role of CH...N hydrogen bond in the formation of wave-like topology in crystals of aza-heterocycles.
S. Kota, A. Vidyasagar, A. Naidu, R. G. Gonnade, K. M. Sureshan*
CrystEngComm., 2012, 14, 519-524.
2011
- Soft optical devices from self-healing gels formed by oil and sugar based organogelators.
A. Vidyasagar, K. Handore, K. M. Sureshan*
Angew. Chem. Int. Ed. 2011, 50, 8021-8024.
Highlighted as Research Highlights in Nature: Nature 2011, 475, 427.
Also Highlighted in Optics & Photonics Focus 2011,15, story 2
Angewandte Highlights Angew. Chem. Int. Ed. 2012, 51, 1760.
Till 2010
- Selective determinants of inositol 1,4,5-trisphosphate and adenophostin A interactions with type-I inositol 1,4,5-trisphosphate receptors.
A. M. Rossi, K. M. Sureshan, A. M. Riley, B. V. L. Potter, C. W. Taylor
British J. Pharmacol. 2010, 161, 1070-1085.
2009
- Total synthesis of cyclitol based natural products from myo-inositol: brahol and pinpollitol.
K. M. Sureshan*, T. Murakami, Y. Watanabe
Tetrahedron 2009, 65, 3998-4006. - Activation of IP3 receptors by synthetic bisphosphate ligands.
K. M. Sureshan, A. M. Riley, A. M. Rossi, S. C. Tovey, S. G. Dedos, C. W. Taylor, B. V. L. Potter
Chem. Commun. 2009, 1204-1206. - Regioselective O-acylation of myo-inositol 1,3,5-orthoesters: Dependence of regioselectivity on the stoichiometry of the base.
K. M. Sureshan, M. S. Shashidhar
Tetrahedron 2009, 65, 2703-2710.
2008
- Efficient syntheses of optically pure chiro- and allo- inositol derivatives, azidocyclitols and aminocyclitols from myo-inositol.
K. M. Sureshan*, K. Ikeda, N. Asano, Y. Watanabe
Tetrahedron 2008, 64, 4072-4080. - Strength from weakness: CH…π stabilized conformational tuning of benzyl ethers and a consequent co-operative edge-to-face CH…π network.
K. M. Sureshan*, T. Uchimaru, Y. Yao, Y. Watanabe
CrystEngComm. 2008, 10, 493-496. - 2-Position base modified analogs of adenophostin A as high affinity agonists of the D-myo-inositol trisphosphate receptor: In vitro evaluation and molecular modeling.
K. M. Sureshan, M. Trusselle, S. C. Tovey, C. W. Taylor, B. V. L. Potter
J. Org. Chem. 2008, 73, 1682-1692.
2007
- Rapid and efficient routes to phosphatidylinositols 3,4,5-trisphosphates via myo-inositol orthobenzoate.
K. M. Sureshan, A. M. Riley, B. V. L. Potter
Tetrahedron Lett. 2007, 48, 1923-1926.
2006
- Guanophostin A: Synthesis and evaluation of a high affinity agonist of the D-myo-inositol 1,4,5-trisphosphate receptor.
K. M. Sureshan, M. Trusselle, S. C. Tovey, C. W. Taylor, B.V. L. Potter
Chem. Commun., 2006, 2015-2017.
2005
- Solid and solution state conformations of (±)-3-O-acetyl-1,2:4,5-di-O-isopropylidene-allo-inositol and (±)-3-O-acetyl-1,2:4,5-di-O-isopropylidene-6-O-methyl-allo-inositol.
K.M. Sureshan*, Y. Watanabe
Carbohydr. Res., 2005, 340, 2311-2318. - Sulfonate protecting groups. Synthesis of O- and C-methylated inositols: D- and L-ononitol, D- and L-laminitol, mytilitol and scyllo-inositol methyl ether.
M. P. Sarmah, M. S. Shashidhar, K. M. Sureshan, R.G. Gonnade, M. M. Bhadbhade
Tetrahedron 2005, 61, 4437-4446. - Establishment of the structure of pinpollitol by total synthesis of the proposed putative structures.
K. M. Sureshan*, T. Murakami, Y. Watanabe
Synlett 2005, 769-772. - Short S=O…C=O contacts associate diastereomers of 2,4(6)-di-O-benzoyl-6(4)-O-[(1S)-10-camphorsulfonyl]-myo-inositol 1,3,5-orthoformates in their inclusion complexes.
K. Manoj, K. M. Sureshan, R. G. Gonnade, M. M. Bhadbhade, M. S. Shashidhar
Cryst. Growth Des. 2005, 5, 833-836. - Resolution of synthetically useful myo-inositol derivatives using the chiral auxiliary O-acetylmandelic acid.
K. M. Sureshan*, Y. Kiyosawa, F. Han, S. Hyodo, Y. Uno, Y. Watanabe
Tetrahedron: Asymmetry 2005, 16, 231-241.
2004
- O-acetylmandelic acid as a reliable chiral anisotropy reagent for the determination of absolute configuration of alcohols.
K.M. Sureshan*, T. Miyasou, S. Miyamori, Y. Watanabe
Tetrahedron: Asymmetry 2004, 15, 3357-3364. - Probing gelation at the molecular level: head-to-tail hydrogen-bonded self-assembly of an inositol-based organogelator.
K. M. Sureshan*, K. Yamaguchi, Y. Sei, Y Watanabe
Eur. J. Org. Chem. 2004, 4703-4709. - Efficient routes to optically active azido-, amino-, diazido- and diaminocyclitols with chiro- and allo- configuration from myo-inositol.
K. M. Sureshan*, K. Ikeda, N. Asano, Y. Watanabe
Tetrahedron Lett., 2004, 45, 8367-8370. - Topochemical transketalization reaction driven by hydrogen bonding.
K. M. Sureshan*, T. Murakami, T. Miyasou, Y. Watanabe
J. Am. Chem. Soc. 2004, 126, 9174-9175. - Solid and solution state conformation of 1L-1-O-acetyl-2,3:5,6-di-O-isopropylidene-chiro-inositol.
K. M. Sureshan*, T. Miyasou, Y. Watanabe
Carbohydr. Res. 2004, 339, 1803-1807. - Crystal structure of 1L-1,2:4,5-di-O-isopropylidene-allo-inositol; A comparison of its conformation in solid and solution states.
K. M. Sureshan*, T. Miyasou, Y. Watanabe
Carbohydr. Res. 2004, 339, 1551-1555. - Total synthesis of the proposed structure of brahol and the structural revision.
K. M. Sureshan, T. Miyasou, Y. Watanabe
Tetrahedron Lett. 2004, 45, 3197-3201. - An efficient route to optically active inositol derivatives via the resolution of myo-inositol 1,3,5-orthoformate: a short synthesis of D-myo-inositol-4-phosphate.
K. M. Sureshan, Y. Watanabe
Tetrahedron: Asymmetry 2004, 15, 1193-1198. - Simple and efficient routes to optically active chiro and allo-inositol derivatives from myo-inositol.
K. M. Sureshan, Y. Watanabe
Synlett. 2004, 493-496. - Crystal structure, solid state and solution conformation of 1D-1,4-di-O-[(S)-O-acetylmandeloyl]2,3:5,6-di-O-isopropylidene-myo-inositol.
K. M. Sureshan*, T. Miyasou, Y. Watanabe
Carbohydr. Res. 2004, 339, 807-811. - Is O-acetylmandelic acid a reliable chiral anisotropy reagent?
K. M. Sureshan, T. Miyaso, M. Hayashi, Y. Watanabe
Tetrahedron: Asymmetry, 2004, 15, 3-7.
2003
- A simple and practical resolution of 1,2:4,5-di-O-isopropylidene-myo-inositol.
K. M. Sureshan, T. Yamasaki, M. Hayashi, Y. Watanabe
Tetrahedron: Asymmetry, 2003, 14, 1771-1774. - Regioselective protection and deprotection of inositol hydroxyl groups.
K. M. Sureshan, M. S. Shashidhar, T. Praveen, T. Das
Chem. Rev. 2003, 103, 4477-4503. - Sulfonate protecting groups: Synthesis of D- and L-myo-inositol-1,3,4,5-tetrakisphosphate precursors by a novel Silver (I) oxide mediated O-alkylation of 2,4(6)-di-O-acyl-6(4)-O-sulfonyl-myo-inositol 1,3,5-orthoformate derivatives through intramolecular assistance of the sulfonyl group.
K. M. Sureshan, T. Das, M. S. Shashidhar, R. G. Gonnade, M. M. Bhadbhade
Eur. J. Org. Chem. 2003, 1035-1041.
2002
- Regioselective sulfonylation of orthoesters of myo-inositol: Formal synthesis of both D- and L-myo-inositol 1,3,4,5 tetrakisphosphate.
K. M. Sureshan, M. S. Shashidhar
Trends Carbohydr. Chem. 2002, 8, 77-86. - Sulfonate protecting groups. Regioselective sulfonylation of myo-inositol orthoesters: Improved synthesis of precursors of D- and L-myo-inositol 1,3,4,5-tetrakisphosphate, myo-inositol-1,3,4,5,6-pentakisphosphate and related derivatives.
K. M. Sureshan, M. S. Shashidhar, T. Praveen, R. G. Gonnade, M. M. Bhadbhade
Carbohydr. Res., 2002, 337, 2399-2410. - Cyclitol based metal complexing agents: Effect of the relative orientation of oxygen atoms in the ionophoric ring on the cation binding ability of myo-inositol based crown ethers.
K. M. Sureshan, M. S. Shashidhar, A. J. Varma
J. Org. Chem., 2002, 67, 6884-6888. - Silver (I) oxide – Silver halide mediated alcoholysis of O-benzoyl-myo-inositol 1,3,5-orthoformates: Intramolecular assistance by the sulfonyl group.
T. Praveen, T. Das, K. M. Sureshan, M. S. Shashidhar, U. Samanta, D. Pal, P. Chakrabarti
J. Chem. Soc. Perkin Trans., 2002, 2, 358-365.
2001
- Neutral complexing agents with a cyclitol core. Effect of the relative orientation of the sidearms and end groups on the cation binding ability of myo-inositol based podands.
K. M. Sureshan, M. S. Shashidhar, A. J. Varma
J. Chem. Soc., Perkin Trans., 2001, 2, 2298–2302. - A highly selective host-guest system formed and stabilized due to concerted halogen-oxygen and C-H ...O non-bonded interactions: X-ray structures of racemic 1,2,3,4,5-penta-O-benzoyl-6-O-tosyl-myo-inositol-dihalomethane (CH2X2, x=Cl and Br) inclusion complexes.
K. M. Sureshan, R. G. Gonnade, M. S. Shashidhar, V. G. Puranik, M. M. Bhadbhade
Chem. Commun., 2001, 881-882. - Sulfonate protecting groups. Regioselective O-sulfonylation of myo-inositol orthoesters.
K. M. Sureshan, M. S. Shashidhar
Tetrahedron Lett., 2001, 42, 3037-3039.
2000
- Regioselective acylation of orthoesters of myo-inositol: causes and consequences.
K. M. Sureshan, M. S. Shashidhar
Trends Carbohydr. Chem., 2000, 6, 59-64. - Regioselective O-acylation of myo-inositol-1,3,5-orthoesters: The role of acyl migration.
K. M. Sureshan, M. S. Shashidhar
Tetrahedron Lett., 2000, 41, 4185-4188l.
No. of Visits 3969

